Research
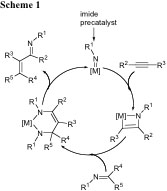
2. Catalytic Reactions Involving Imide and Phosphinidene Group Transfer Chemistry.
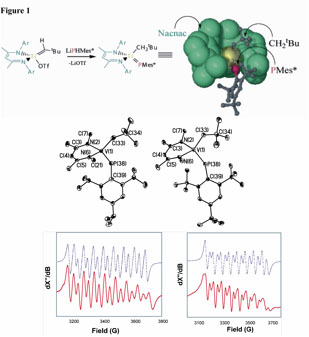
Catalysts increment reaction rates by reducing the activation barrier of the reaction, yet remain unchanged upon completion of one catalytic cycle. Depending of the efficacy of the catalyst, energy expenditures can be ultimately reduced, and the amount of product can be conveniently produced for subsequent research such as the synthesis of fine chemicals or just simply for use as a commodity product. In some cases however, the nature of the reaction or catalyst allow for atom-efficient systems, where waste (mostly by-products) are minimized with much cleaner, catalytic alternatives. Our group is interested in catalytic C-N, C-C and C-P forming reactions, since the products derived from these types of transformations offer the opportunity to study these systems from both an applied and fundamental point of view. Specifically, we are focused on the formation of α,β-unsaturated imine frameworks since this is a new class of highly arylated, organic archetypes. These types of frameworks are interesting given the highly conjugated nature surrounding the ene-imine functionality as well as the mechanism to their formation, since C=C and C=N bonds are catalytically formed while C-N bonds are catalytically broken (Scheme 1). Our group has developed efficient catalysts for carboamination1 reactions of PhCCPh and aldimines to produce highly arylated α,β-unsaturated imines with exclusive (E,E)-configuration at the olefin and imine residues.2,3 We are currently exploring C-N and C-P forming reactions such as hydroamination and hydrophosphination using pre-catalysts having terminal M=NR and M=PR linkages. We are also exploring the catalytic activity (hydrophosphination) of terminal Ti and V phosphinidene complexes prepared by our group (Figure 1 depicts the first vanadium and titanium complexes bearing a terminal phosphinidene scaffold).
Selected References
1. Ruck, R. T.; Zuckermann, R. L.; Krska, S. W.; Bergman, R. G. Angew. Chem. Int. Ed. 2004, 43, 5372-5374.
2. Basuli, F.; Aneetha, H.; Huffman, J. C.; Mindiola, D. J. J. Am. Chem. Soc. 2005, 127, 17992-17993.
3. Aneetha, H.; Basuli, F.; Huffman, J. C.; Mindiola, D. J. Organometallics 2006, 25, 2402-2404.
