Research
5. Modeling the Hydrodenitrogenation (HDN) Process.
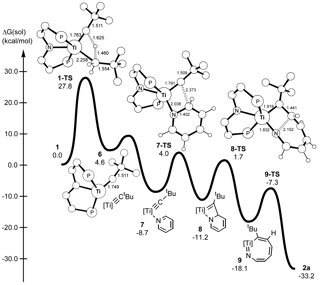
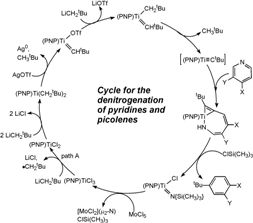
Extrusion of nitrogen as ammonia from organic compounds constitutes an important transformation, and it is crucial to widely utilized industrial applications such as hydrodenitrogenation of crude oil, as well as the catalytic degradation of heterocycles. Removal of nitrogen-containing impurities typically involves treatment of crude oils with high pressures of hydrogen (150-2250 psi) and high temperatures (350-500 °C) over a heterogeneous catalyst (typically a sulfided CoMo/γ-Al2O3 or NiMo/γ-Al2O3 surface). If the heteroatom is not removed during the process, it can lead to the release of NOx gases upon fuel combustion and contribute to the formation of acid rain. The N-heterocycle can also be a poison, thus interfering with other important processes such as reforming catalysts as well as hydrocracking. We are interested in examining the role of metal in the binding of heterocycles, and ultimately exploring the mechanism leading to rupture of the strong aromatic N-C bond. In this respect, cleavage of the carbon-nitrogen bond of pyridine, an N-heterocycle of remarkable stability, is difficult, and only a few well-defined homogeneous examples of this potentially useful transformation have been documented.1-3 This chemical paradigm is particularly attractive given its relevance to important industrial processes such as hydrodenitrogenation (HDN); a reaction in which N-heterocycles present in petroleum or coal-based liquids are catalytically converted to NH3 and nitrogen-free carbon-hydrogen based products. As a result, understanding the mechanism by which strong C-N bonds of aromatic substrates such as pyridine can be cleaved offer an excellent opportunity to improve or develop more efficient HDN catalysts. Our group has discovered the facile and non-reductive C-N activation and cleavage of pyridine by a transient titanium alkylidyne (PNP)Ti≡CtBu (PNP = N[2-P(CHMe2)2-4-methylphenyl]2-). We have determined that activation of the C-N bond in pyridine involves a unique process; a room temperature ring-opening metathesis (ROM) step invoking the [2+2] cycloaddition of the aromatic C=N bond across a reactive Ti≡CtBu linkage.4 The reaction profile for this rare phenomenon is depicted to the right. Using a combination of synthesis, isotopic labeling, and theoretical studies we are now beginning to understand a unique transformation in which the aromatic ring of the N-heterocycle is cleaved at room temperature! Our group has also found that the nitrogen composing the ring-opened product in 2a, can be denitrogenated, under mild conditions (albeit slowly), when using an electrophile such as R3SiCl.5 The scheme show depicts a cyclic, and homogeneous denitrogenation reaction of pyridine, without even using hydrogen for hydrogenolysis of the C-N bonds. The titanium imide product, can be recycled using two equivalents of MoCl5. Subsequent steps can then yield the alkylidyne synthon.
Selected References
1. Kleckley, T. S.; Bennett, J. L.; Wolczanski, P. T.; Lobkovsky, E. B. J. Am. Chem. Soc. 1997,
119, 247-248.
2. (a) Strickler, J. R.; Bruck, M. A.; Wigley, D. A. J. Am. Chem. Soc. 1990,
112, 2814-2816. (b) Gray, S. D.; Weller, K. J.; Bruck, M. A.; Briggs, P. M.; Wigley, D. E.
J. Am. Chem. Soc. 1995, 117, 10678-10693. (c) Gray, S. D.; Smith, D. P.; Bruck, M. A.;
Wigley, D. E. J. Am. Chem. Soc. 1992, 114, 5462-5463.
3. Laine, R. M.; Thomas, D. W.; Cary, L. W. J. Org. Chem. 1979, 44, 4964-4966.
4. Bailey, B. C.; Fan, H.; Huffman, J. C.; Baik, M.-H.; Mindiola, D. J. J. Am. Chem. Soc.
2006, 128, 6798-6799.
5. Fout, A. F.; Bailey, B. C.; Tomaszewski, J.; Mindiola, D. J. J. Am. Chem. Soc. 2007, 129,
12640-12641.
